|
8/11/2023 0 Comments C2h2 vsepr 
With fewer 90° LP–BP repulsions, we can predict that the structure with the lone pair of electrons in the equatorial position is more stable than the one with the lone pair in the axial position. Looking at the C2H2 Lewis structure we can see that there are two atoms attached to the Carbon of interest and that there no lone pair electrons. Question: Using Lewis Structures and the VSEPR model, predict the molecular geometries of CO2, CH4, C2H2,C2H4, and C2H6 and then use the geometries to describe the hybridization (sp, sp2, or sp3) expected for each carbon atom. Its odor is described as faintly ‘sweet and musky’. 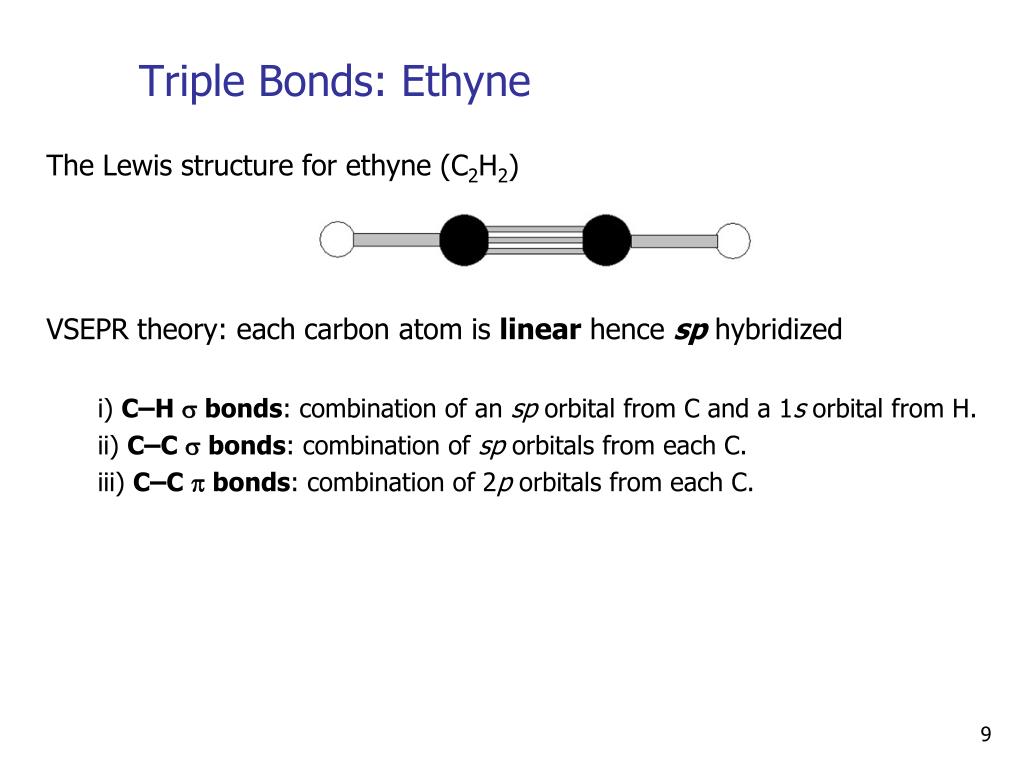
C2H4 exists as a colorless gas and is flammable. An alkene is a hydrocarbon with a Carbon-Carbon double bond. This molecule is also represented by H2CCH2, clearly showing the alkene nature of the compound. O linear bent trigonal-planar tetrahedral octahedral. 16 Jul The chemical formula C2H4 represents Ethylene. Question: Use VSEPR theory to predict the molecular geometry around either carbon atom in acetylene, C2H2. Ethyne has a triple bond between the two carbon atoms. Youll get a detailed solution from a subject matter expert that helps you learn core concepts. The number of electrons on the valence shell of Be and Cl is 2. The electronic configuration of beryllium is He 2s2and chlorine is Ne 3s23p5. These electrons will be both bonding as well as non-bonding electrons. for the peripheral atoms in a molecule and apply the VSEPR model to these. The 2s orbital in each carbon hybridizes with one of the 2p orbitals and forms two sp hybrid orbitals. The electrons present in the outermost shell of an atom are shown in the Lewis structure of any molecule. hybridization is acetylene (C2H2 ), which has the systematic name ethyne. As a result, one 2s 2 pair is moved to the empty 2pz orbital. If we place it in the axial position, we have two 90° LP–BP repulsions at 90°. In the formation of C2H2, the carbon atom needs extra electrons to form 4 bonds with hydrogen and other carbon atoms. However, because the axial and equatorial positions are not chemically equivalent, where do we place the lone pair? If we place the lone pair in the equatorial position, we have three LP–BP repulsions at 90°. VSEPR Theory - Acetylene (C2H2) Professor Heath's Chemistry Channel 18.2K subscribers Subscribe 37 Share 13K views 10 years ago Acetylene For the molecule acetylene (C2H2): a) Draw the Lewis. We designate SF 4 as AX 4E it has a total of five electron pairs. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
